EARLY DETECTION
Pancreatic cancer: the case for catching it early

Written by
Nick Lenten

Reviewed by

Pancreatic cancer often progresses undetected for a long time – and is therefore frequently diagnosed late. In Switzerland, around 1,500 people are diagnosed each year. Only about 12% survive five years.
There is no standard screening test. Symptoms blend into everyday discomfort. By the time a diagnosis arrives, the cancer has usually already escaped the pancreas.
That picture is starting to shift. Full-body MRI can now detect pancreatic abnormalities before any symptoms appear, giving people – particularly those with elevated risk – a way to act before the disease takes the upper hand.
What is pancreatic cancer?
The pancreas sits behind the stomach and has two major functions: produces enzymes to digest food, and releases hormones like insulin to regulate blood sugar. Cancer begins when cells in the organ multiply without restraint, forming a malignant tumour.
About 90% of cases are pancreatic ductal adenocarcinoma (PDAC), beginning in the cells lining the pancreatic ducts. The organ's position makes detection genuinely difficult. Tucked deep in the abdomen, shielded by other organs and major blood vessels, the pancreas hides tumours well. They grow silently – producing no symptoms until they have invaded surrounding tissue or spread to distant sites.
Survival rates: the numbers tell the story
The 5-year survival rate is between 9% and 13% across all stages
In Switzerland, roughly 12% of patients survive five years post-diagnosis
Surgery – the only potentially curative treatment – is available to just 15–20% of patients at diagnosis; the remaining 80–85% arrive too late
The average age at diagnosis is approximately 70–75 years, though younger people are not immune
These numbers reflect late detection, not untreatable biology. When pancreatic cancer is caught at Stage I, still confined to the organ, survival improves dramatically. Early detection is the variable that determines who lives and who does not.
Signs and symptoms: what to watch for
Pancreatic cancer imitates common, harmless conditions in its early stages. Many patients wait weeks or months before seeing a doctor.
Upper abdominal pain or back pain: a dull, persistent and often belt-like ache in the upper abdomen, often radiating to the back, worse after eating or lying down.
Jaundice (yellowing of the skin and eyes): caused by a tumour blocking the bile duct, often accompanied by dark urine, pale stools, and itchy skin.
Unexplained weight loss: significant loss without dietary or exercise changes.
Loss of appetite: sudden, persistent disinterest in food.
Steatorrhoea: fatty, light-coloured, foul-smelling stools, caused by the pancreas failing to produce enough digestive enzymes.
New-onset diabetes: a sudden diabetes diagnosis, especially in someone without typical risk factors, can signal pancreatic cancer. An estimated 5–10% of diabetics in industrialised nations have Type 3c diabetes, caused by pancreatic disease rather than metabolic syndrome.
Fatigue: persistent exhaustion unrelieved by rest.
Any combination of these symptoms – particularly new-onset diabetes alongside unexplained weight loss or abdominal pain – warrants prompt medical evaluation. The gap between a curable and incurable diagnosis can be a matter of weeks.
Risk factors
Smoking
The single biggest modifiable risk factor. Smokers face roughly twice the risk of developing pancreatic cancer compared to non-smokers, with some studies citing an 80% increase. Tobacco carcinogens damage pancreatic cells cumulatively over years.
Alcohol consumption
More than 30 grams of alcohol per day (roughly two beers or three glasses of wine) is linked to a moderate increase in risk. Heavy, prolonged drinking also causes chronic pancreatitis, itself a risk factor.
Overweight and obesity
An estimated 15–18% of pancreatic carcinomas are attributable to excess weight, through mechanisms of chronic inflammation and insulin resistance.
Type 2 diabetes
Long-standing type 2 diabetes (more than three years) raises pancreatic cancer risk 1.5 to 2 times. The relationship runs both ways: diabetes can cause and be caused by pancreatic cancer.
Chronic pancreatitis
Chronic inflammation of the pancreas increases risk 2 to 6 times depending on duration and cause.
Family history and genetics
About 8% of pancreatic cancer cases have a family history component
BRCA1 mutation carriers face a 2 to 3 times higher risk than the general population
Other hereditary conditions linked to elevated risk include BRCA2, Lynch syndrome, familial atypical multiple mole melanoma (FAMMM) syndrome, and familial adenomatous polyposis (FAP)
Age
Risk rises sharply after 60. Most diagnoses occur in people over 65.
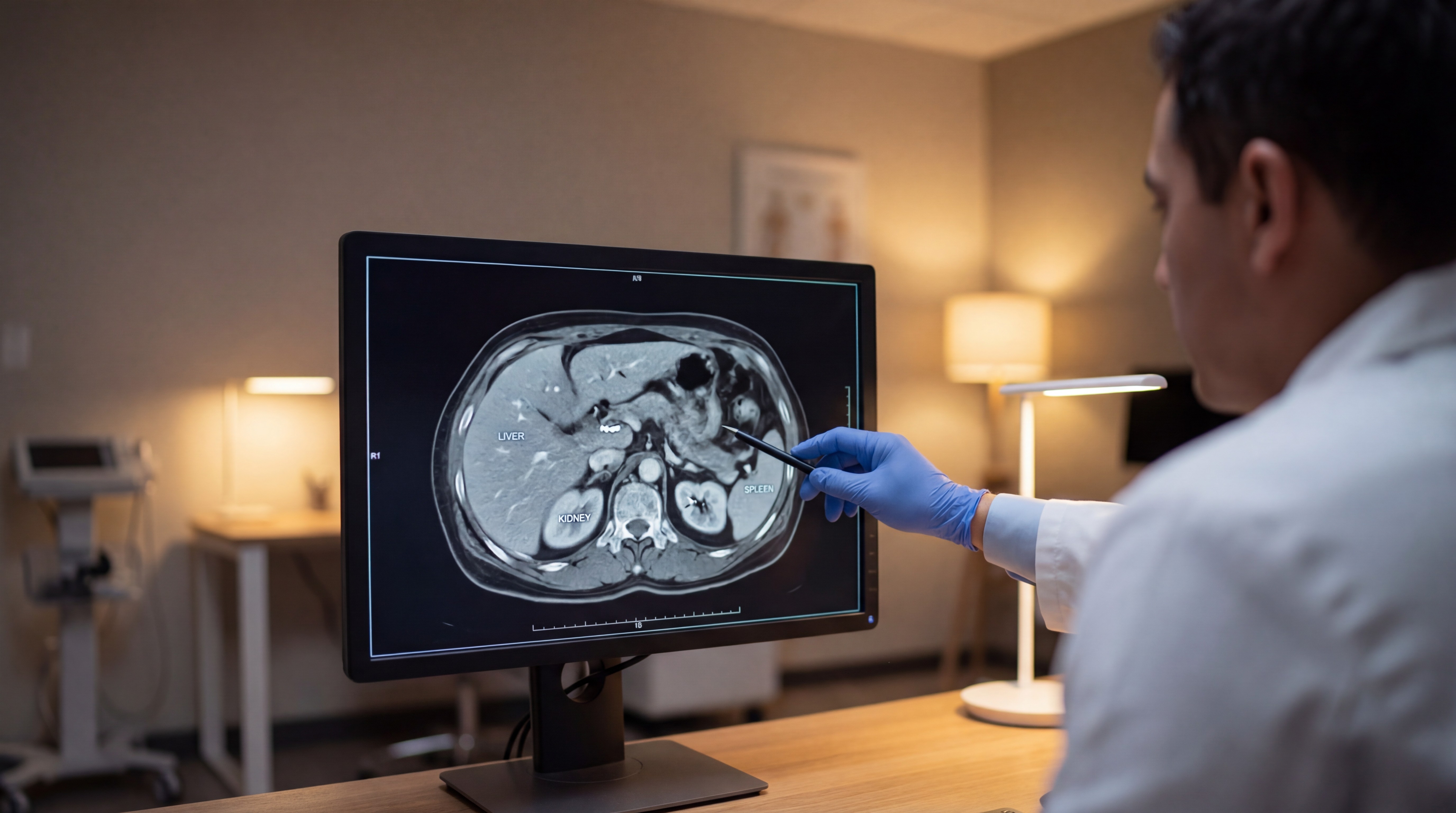
Diagnosis
There is no population-wide screening programme for pancreatic cancer. Diagnosis typically begins only after symptoms appear.
Imaging
Abdominal ultrasound: usually the first test. Non-invasive, but limited in visualising small tumours buried deep in the pancreas.
CT scan: the workhorse for staging pancreatic cancer, providing detailed cross-sectional images.
MRI: high-resolution, radiation-free soft-tissue imaging, especially useful for spotting small lesions and mapping a tumour's relationship to surrounding structures. Full-body MRI can detect pancreatic abnormalities before they cause symptoms.
PET scan: a radioactive tracer highlights metabolically active cancer cells, revealing whether the disease has spread.
Blood and laboratory tests
CA 19-9 is the most widely used tumour marker for pancreatic cancer. It is not definitive alone, but elevated levels support a diagnosis and help track treatment response. Standard blood panels covering liver function and blood sugar can also flag pancreatic dysfunction.
Biopsy
A tissue sample from the suspected tumour, examined under a microscope. The only definitive confirmation of pancreatic cancer.
Genetic testing
For patients with family history or known hereditary cancer syndromes, testing can identify mutations in BRCA1, BRCA2, PALB2, and other genes. Results guide both surveillance and treatment.
Treatment
What is possible depends entirely on timing.
Surgery
The only potentially curative treatment, feasible only when the tumour is localised and has not invaded major blood vessels. The most common procedure is the Whipple procedure (pancreatoduodenectomy), removing the head of the pancreas along with parts of the small intestine, bile duct, and sometimes the stomach. Only 15–20% of patients qualify at diagnosis.
Chemotherapy
Used before surgery (neoadjuvant) to shrink the tumour, after surgery (adjuvant) to destroy remaining cells, and palliatively in advanced cases to slow progression and improve quality of life.
Radiotherapy
Often paired with chemotherapy (chemoradiation), particularly for locally advanced cancers that cannot be removed surgically.
Palliative care
When cure is not possible, palliative care manages pain, jaundice, digestive problems, and nutrition to preserve quality of life.
What is changing in pancreatic cancer research?
Several lines of research are gaining meaningful ground.
Better imaging. High-resolution MRI and AI-enhanced image analysis are catching smaller lesions and pre-cancerous changes earlier than was possible even five years ago.
Liquid biopsies. Blood tests that detect circulating tumour DNA could identify pancreatic cancer before imaging reveals a visible mass.
Targeted therapies. KRAS mutations appear in over 90% of pancreatic cancers. Researchers are developing drugs that target these mutations directly – a potential shift in how the disease is treated, not just managed.
Immunotherapy. Pancreatic cancer has historically resisted immune checkpoint inhibitors, but combinations with other treatments are showing early promise in clinical trials.
AI diagnostics. Machine-learning models trained on imaging data are identifying pancreatic abnormalities that human radiologists sometimes miss.
All of these advances share a common thread: the earlier the cancer is found, the more they can offer.
Why detection beats treatment
Every pancreatic cancer statistic circles back to timing. When found at Stage I, survival rates climb sharply. When found at Stage III or IV, options narrow fast.
Standard Swiss care is reactive. You see a doctor when something feels wrong. With pancreatic cancer, by the time something feels wrong, the window for curative treatment has often already closed. This is the asymmetry that preventive screening addresses.
How Ahead Health supports early detection
Ahead Health works on a different principle: find the problem before it announces itself.
Ahead's full-body MRI delivers radiation-free imaging of the entire body, including the pancreas and surrounding abdominal organs. Unlike standard check-ups, it can reveal pancreatic cysts and lesions that may be pre-cancerous, structural abnormalities in the pancreas, bile ducts, and surrounding tissues, and masses too small to cause symptoms but significant enough to warrant monitoring. You can see the full list of conditions Ahead screens for on the what we detect page.
The Ahead Core package (CHF 1,990) provides the full-body MRI and a health report reviewed by Swiss board-certified physicians – a practical starting point for anyone wanting a whole-body baseline. The Ahead Advanced package (CHF 2,490) adds an 80+ biomarker blood panel, covering metabolic markers and organ function indicators relevant to pancreatic health, including blood glucose, HbA1c, and liver enzymes that can signal early pancreatic dysfunction.
Every scan is supported by AI-assisted analysis, helping the medical team identify subtle patterns with greater precision. As Dr. Anna Erat, Ahead's founding medical advisor, puts it: "With pancreatic cancer, the right questions are structural and metabolic: what does the scan show, and what are the markers telling us? We can answer both of those years before any symptoms appear." Imaging alone tells part of the story; combined with blood data, the picture becomes substantially more complete.
Screening is especially important if you carry one or more elevated risk factors: family history of pancreatic cancer, BRCA1 or BRCA2 mutations, chronic pancreatitis, long-standing type 2 diabetes, a smoking history, or age over 50.
Even without specific risk factors, a full-body MRI establishes a baseline – a reference against which future scans can detect even subtle changes over time.
Supplementary health insurers may cover part of the cost. KPT, CSS, AXA, Visana and Atupri are all reimbursing part of the cost for Ahead Health services depending on their T&Cs, some with up to CHF 1,500. You can check how much you can get reimbursed and request a pro-forma invoice on our insurance page.
Conclusion
Pancreatic cancer's grim statistics are largely a story of late detection. The biology is not uniquely intractable. The problem is that by the time symptoms appear, the window for curative treatment has usually passed. Full-body MRI changes that equation – catching structural changes before they become emergencies. For people with elevated risk factors, proactive screening is the most concrete step available.
Ahead Health is a Swiss preventive healthcare company offering full-body MRI scans, advanced blood tests, and AI-powered health insights. Learn more at aheadhealth.com.
FAQ
What is the survival rate for pancreatic cancer?
What are the earliest symptoms of pancreatic cancer?
Is there a cure for pancreatic cancer?